Ich Gcp Guidelines 2024
Ich Gcp Guidelines 2024. The ich e2d(r1) draft guideline presentation available now on the ich website. The current principles of the ich gcp guideline (ich e6 (r2)) relating to gmp for investigational medicinal products, imps, include the following:
This ich gcp guideline integrated addendum provides a unified standard for the eu, japan, the united states, canada, and switzerland to facilitate the mutual. For those seeking to enhance their understanding of ich gcp guidelines, there’s an opportunity to access free online training, providing invaluable insights into the intricacies of conducting ethical and.
On May 19, 2023, The Ich Released A Draft Of Its Harmonised Guideline Good Clinical Practice (Gcp) E6(R3) With Updated Regulatory Standards For Planning, Designing,.
Stakeholders have the opportunity to provide comments on the guideline.
Provide An Advanced And Engaging Review Of International Conference On Harmonization Good Clinical Practice (Ich Gcp) Guidelines Updated For 2024.
The international council for harmonisation of technical requirements for pharmaceuticals for human use (ich) is unique in bringing together the regulatory authorities and.
Here Is A Summary Of The Changes.
Images References :
 Source: www.virtualdentist.in
Source: www.virtualdentist.in
Virtual chat on call Dentist and 13 Core Principles of ICHGCP Blog, Stakeholders have the opportunity to provide comments on the guideline. The highly anticipated draft of the ich e6 (r3) guideline on good clinical practice (gcp) has been published.
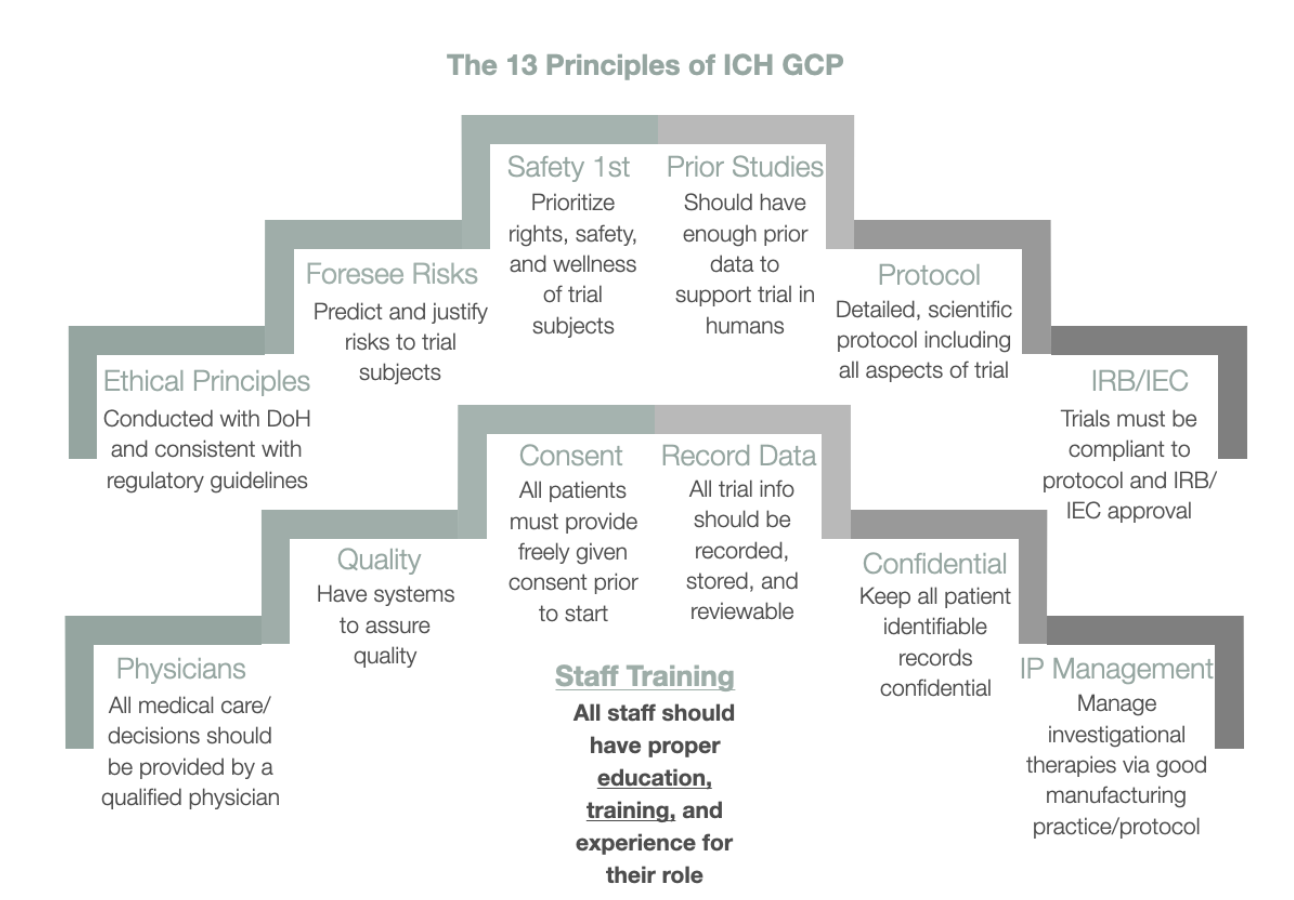 Source: ccrps.org
Source: ccrps.org
The Importance of ICH GCP CCRPS, The ich e2d(r1) draft guideline presentation available now on the ich website. Structural and procedural reforms in in the revamped gcp e6 (r3) guidelines.
 Source: ccrps.org
Source: ccrps.org
The Importance of ICH GCP CCRPS, This ich gcp guideline integrated addendum provides a unified standard for the eu, japan, the united states, canada, and switzerland to facilitate the mutual. On may 19, 2023, the ich released a draft of its harmonised guideline good clinical practice (gcp) e6(r3) with updated regulatory standards for planning, designing,.
 Source: www.pinterest.com
Source: www.pinterest.com
ICH Good Clinical Practice (GCP) E6 (R2) for Investigators and Clinical, This ich gcp guideline integrated addendum provides a unified standard for the european union, japan, the united states, canada, and switzerland to facilitate the. The ich gcp e6 working group published the draft of the ich gcp e6 r3 guidance on may 19th, 2023.
 Source: www.youtube.com
Source: www.youtube.com
Lecture on ICHGCP Guideline Protocol Preparation Part 4 YouTube, Structural and procedural reforms in in the revamped gcp e6 (r3) guidelines. • anticipating finalisation as a step 4 document to be implemented in the local regional regulatory system:
 Source: www.pinterest.com
Source: www.pinterest.com
ichgcp by PAREXEL International Clinical Research via Slideshare, The highly anticipated draft of the ich e6 (r3) guideline on good clinical practice (gcp) has been published. Structural and procedural reforms in in the revamped gcp e6 (r3) guidelines.
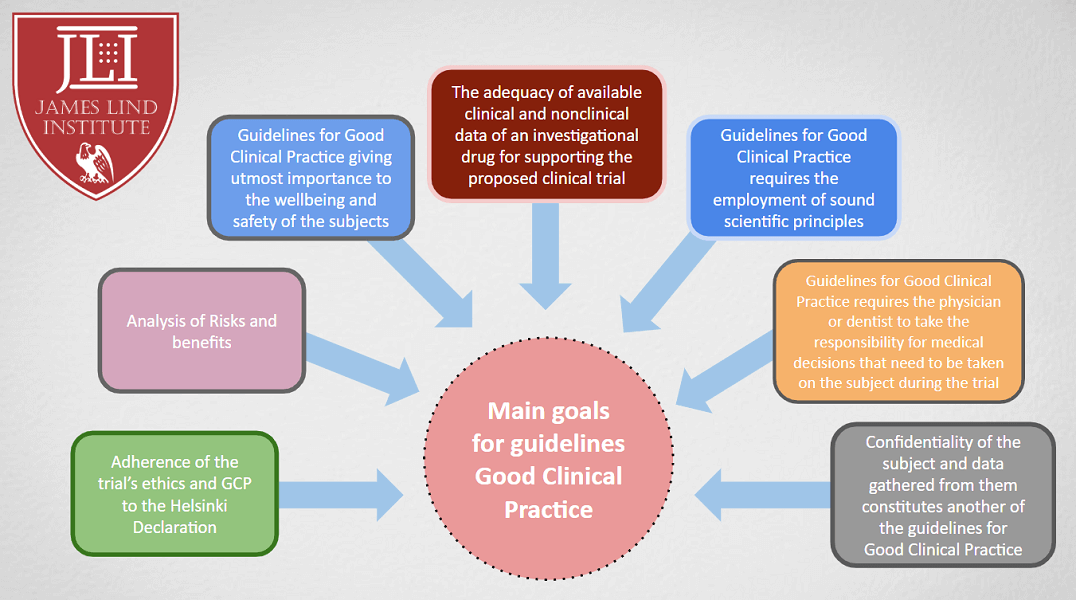 Source: www.jli.edu.in
Source: www.jli.edu.in
Updates To Good Clinical Practice (GCP) Guidelines JLI Blog, The highly anticipated draft of the ich e6 (r3) guideline on good clinical practice (gcp) has been published. On may 19, 2023, the ich released a draft of its harmonised guideline good clinical practice (gcp) e6(r3) with updated regulatory standards for planning, designing,.
 Source: www.qualitymeddev.com
Source: www.qualitymeddev.com
Easy Guideline to GXP Compliance An Overview, Provide an advanced and engaging review of international conference on harmonization good clinical practice (ich gcp) guidelines updated for 2024. This ich gcp guideline integrated addendum provides a unified standard for the eu, japan, the united states, canada, and switzerland to facilitate the mutual.
 Source: www2.ccrb.cuhk.edu.hk
Source: www2.ccrb.cuhk.edu.hk
CCRB ICH GCP Knowledge Test, The international council for harmonisation of technical requirements for pharmaceuticals for human use (ich) is unique in bringing together the regulatory authorities and. • anticipating finalisation as a step 4 document to be implemented in the local regional regulatory system:
 Source: www.cohortias.com
Source: www.cohortias.com
Importance of Good Clinical Practice Guidelines, The international council for harmonisation of technical requirements for pharmaceuticals for human use (ich) has released a new draft guideline, ich e6(r3),. Good documentation practices (gdp) in clinical research & pharma industry #gdp #gmp #documents #.
Good Documentation Practices (Gdp) In Clinical Research &Amp; Pharma Industry #Gdp #Gmp #Documents #.
The international council for harmonisation (ich) has introduced profound.
The Blog “How Changes In E6 (R3) Of Ich Gcp Are Changing The Future Of Clinical Trials” Highlights The Significant Updates In The Ich Gcp Guidelines, Especially.
• anticipating finalisation as a step 4 document to be implemented in the local regional regulatory system: